 2DG
2DG
COVID-19: Dr. Reddy’s Laboratories announces commercial launch of 2DG
Hyderabad: Dr. Reddy’s Laboratories Ltd. on Monday announced the commercial launch of 2-deoxy-D-glucose (2-DG).
Dr. Reddy’s will supply to major Government as well as private hospitals across India.
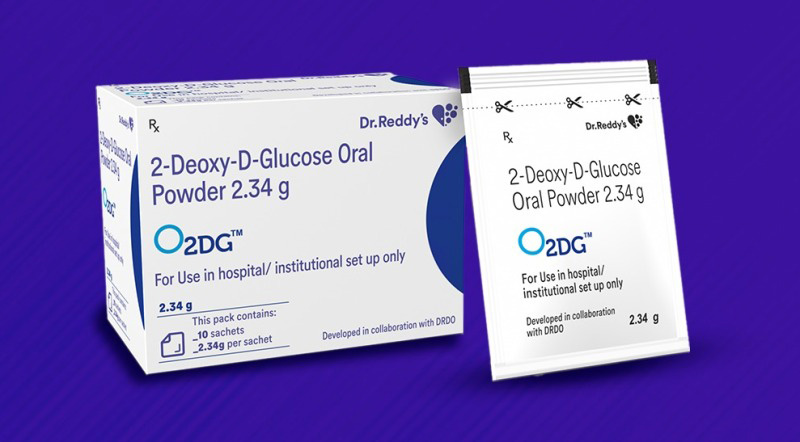
In the initial weeks, the Company will make the drug available in hospitals across metros and Tier 1 cities, and subsequently expand coverage to the rest of India. 2-DG manufactured by Dr. Reddy’s has a purity of 99.5% and is being sold commercially under the brand name 2DGTM.
"The maximum retail price (MRP) of each sachet has been fixed at Rs. 990, with a subsidized rate offered to Government institutions. Enquiries for 2DGTM can be sent to 2DG@drreddys.com. 2-DG was developed by the Institute of Nuclear Medicine & Allied Sciences (INMAS), a laboratory of the Defence Research and Development Organisation (DRDO), in collaboration with Dr. Reddy’s," Dr. Reddy's said in a statement.
2-DG is an oral drug. It can be administered only upon prescription and under the supervision of a qualified physician to hospitalised moderate to severe COVID-19 patients as an adjunct therapy to the existing standard of care.
Emergency use approval for anti-COVID-19 therapeutic application of the drug was granted on May 1, 2021.
Dr. G. Satheesh Reddy, Secretary Department of Defence (R&D) and Chairman, DRDO said: “We are pleased to have worked closely with our long-term industry partner Dr. Reddy's Laboratories, Hyderabad, for testing 2-DG as therapeutic application in treatment of COVID-19 patients. DRDO has been contributing in fight against COVID-19 pandemic with its spin off technologies."
Satish Reddy, Chairman, Dr. Reddy’s said: “2-DG is yet another addition to our COVID-19 portfolio that already covers the full spectrum of mild to moderate and severe conditions and includes a vaccine. We are extremely pleased to have partnered with DRDO in our collective fight against the COVID-19 pandemic.”
Support Our Journalism
We cannot do without you.. your contribution supports unbiased journalism
IBNS is not driven by any ism- not wokeism, not racism, not skewed secularism, not hyper right-wing or left liberal ideals, nor by any hardline religious beliefs or hyper nationalism. We want to serve you good old objective news, as they are. We do not judge or preach. We let people decide for themselves. We only try to present factual and well-sourced news.







